 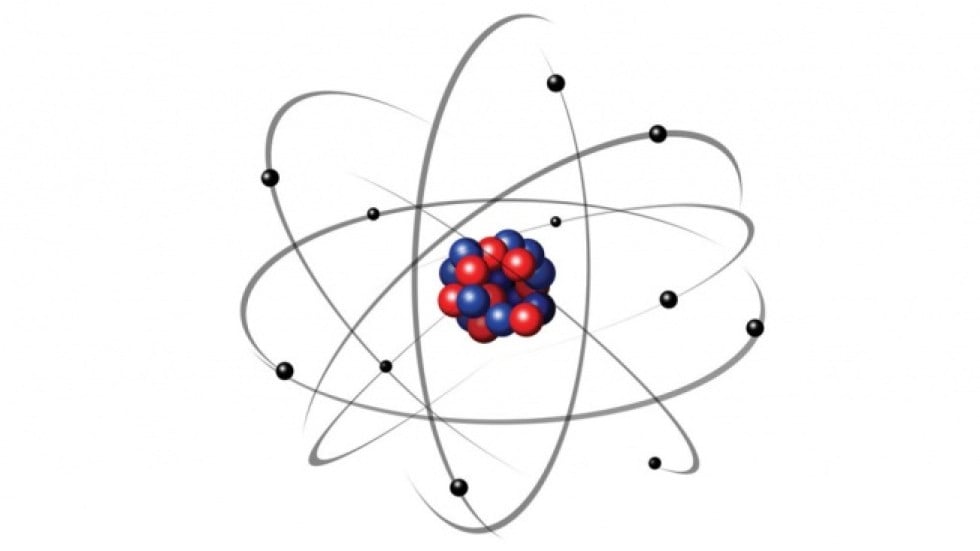
The energy levels of electrons at different orbits are represented by n= 1, 2, 3…. These electrons have definite circular paths known as orbits or shells.Įach of the circular paths has fixed levels of energy and are called orbital shells in terms of this atomic model. Negatively charged electrons revolve around a positively charged nucleus in an atom. The following are the chief characteristics of the atomic model given by Niels Bohr. It was hence proposed that an electron can jump in-between these orbits by gaining or releasing photons (energy). 
Now, an atom that is in the state of the smallest orbit will be completely stable as the electron will not have a lower orbit it can jump to. The reason for atoms being unstable is that electrons jump to lower orbits causing radiation. Therefore, smaller orbits will have lower energy electrons. To describe Bohr's atomic model, it can be said that the energy levels of the electrons concentrate on the size of the orbits. Niels Bohr atomic theory featured fixed size and energy of the electrons moving in orbits around the nucleus which is charged positively similar to the motion of planets around the sun. Bohr's model was backed up by the classical law of physics and the quantum theory of radiation. This model provided a remedy for the stability problem under the previous Rutherford model of the atom, which displayed a motion where electrons would lose their energy and subsequently spiral into the nucleus due to the emission of electromagnetic radiation by the charged particles. A quantized shell model of an atom was given by Bohr in 1913 to provide an explanation of how electrons can stably orbit around the nucleus. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
